Bonding Characteristics in Some Sustainable Water-Soluble Sulfonated PEPPSI–Pd–NHC Catalysts
Keywords:
Pd–NHC Complexes , Sustainable Catalysis , Density Functional Theory (DFT), Natural Bond Orbital Analysis (NBO)Abstract
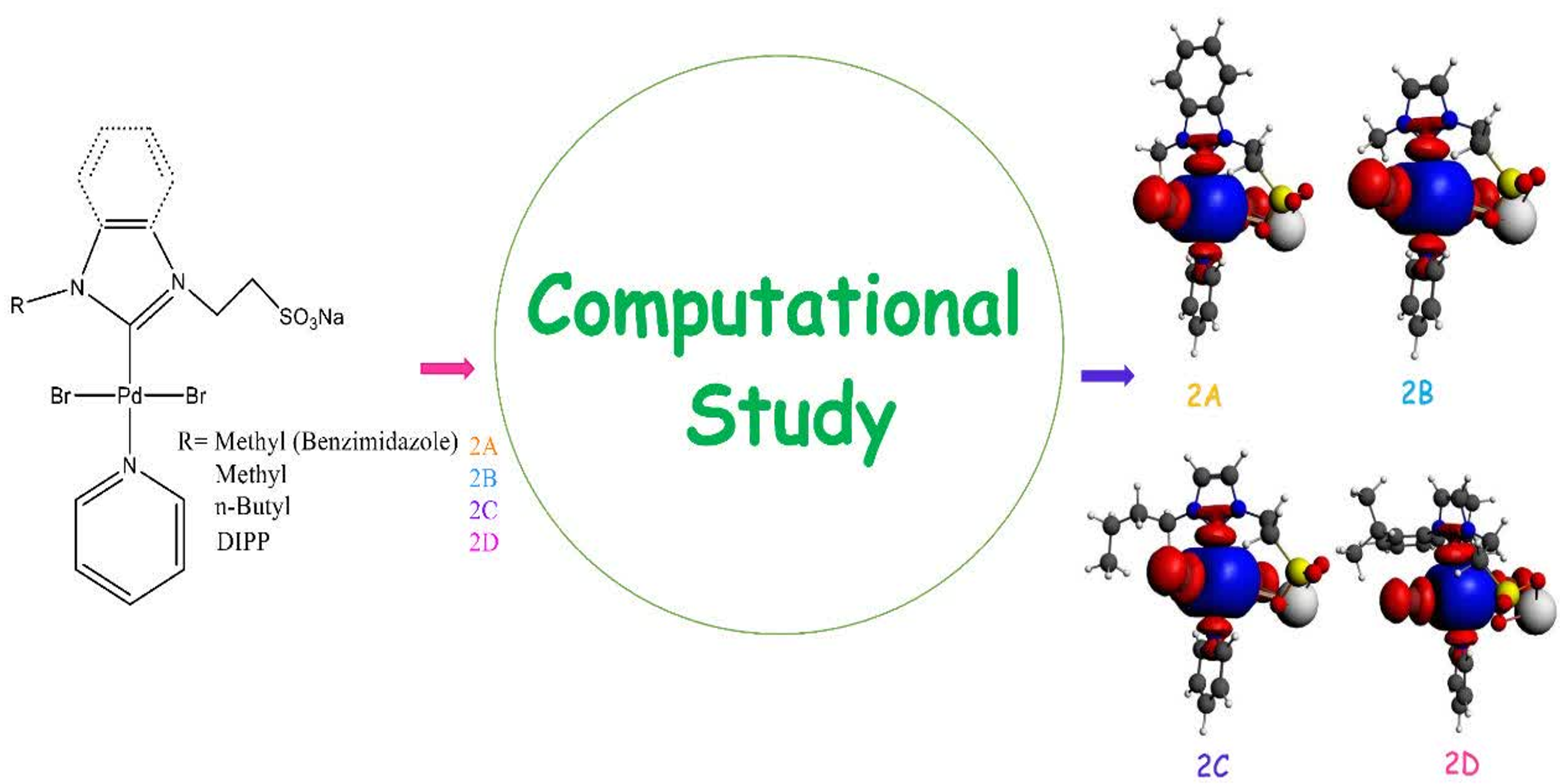
A density functional theory (DFT) study was performed to investigate the structural and electronic properties of four sulfonated, water-soluble PEPPSI–Pd–NHC complexes (2a–2d). All complexes were fully optimized, followed by higher-level single-point calculations to obtain an accurate description of their electronic structures. Metal–ligand interaction energies were evaluated to assess the strength and stability of the Pd–NHC bonding. Natural Bond Orbital (NBO) and Energy Decomposition Analysis (EDA) were employed to analyze charge distribution, bonding characteristics, and the nature of Pd–ligand interactions. The results reveal that variations in the NHC ligands significantly influence the electronic environment of the Palladium center. Notably, the catalyst bearing a 2,6-diisopropylphenyl substituent exhibits the strongest metal–ligand interaction, in good agreement with its superior experimental performance
Downloads
References
N. Miyaura & A. Suzuki, (1995). Chemical Reviews, 95(7), 2457–2483.
E. I. Negishi, (2010). Angewandte Chemie International Edition, 50(30), 6738–6764.
F. Diederich & P. J. Stang (Eds.). (2008). Metal-catalyzed cross-coupling reactions. John Wiley & Sons.
M. Aksoy, H. Kilic, B. Nişancı & Ö. Metin, (2021). Inorganic Chemistry Frontiers, 8(2), 499-545.
J. Meurig Thomas & R. Raja, (2005). Rev. Mater. Res., 35(1), 315-350.
Q. Chen, L. Lv, M. Yu, Y. Shi, Y. Li, G. Pang, & C. Cao, (2013). RSC Advances, 3(40), 18359-18366.
A. J. Arduengo, R. L. Harlow & M. Kline (1991). Journal of the American Chemical Society, 113(1), 361–363.
W. A. Herrmann (2002). Angewandte Chemie International Edition, 41(8), 1290–1309.
S. P. Nolan (2014). N-Heterocyclic Carbenes: Effective Tools for Organometallic Synthesis. Wiley-VCH.
F. Glorius (2007). N-Heterocyclic Carbenes in Transition Metal Catalysis. Springer.
M. G. Organ, G. A., Chass, D. C. Fang, A.C. Hopkinson & C. Valente, (2008). Synthesis, 18, 2776–2797.
C. Valente, S. Çalimsiz, K. H. Hoi, D. Mallik, M. Sayah & M.G. Organ (2012). Angewandte Chemie International Edition, 51(14), 3314–3332.
N. Marion & S. P. Nolan (2008). Accounts of Chemical Research, 41(11), 1440–1449.
F. Mazars, G. Zaragoza & L. Delaude (2023). Inorganica Chimica Acta, 556, 121676.
N. Hamdi, I. Slimani, L. Mansour, F. Alresheedi, N. Gürbüz & I. Özdemir, (2021). New Journal of Chemistry, 45(45), 21248-21262.
G. Frenking & F. M. Bickelhaupt (2014). Chemical Society Reviews, 43(2), 510–530.
A. V. Astakhov, S. B. Soliev & V. M. Chernyshev, (2020). Russian Chemical Bulletin, 69(11), 2073-2081.
W. Koch & M. C. Holthausen, (2001). A Chemist’s Guide to Density Functional Theory. Wiley-VCH.
F. Jensen, (2017). Introduction to computational chemistry. John wiley & sons.
C. J. Cramer, (2013). Essentials of Computational Chemistry. Wiley.
A. E. Reed, R. B. Weinstock & F. Weinhold, (1985). Journal of Chemical Physics, 83(2), 735–746.
F. Weinhold & C. R. Landis (2005). Valency and Bonding. Cambridge University Press.
A. Krapp, F. M. Bickelhaupt & G. Frenking, (2006). Chemistry–A European Journal, 12(36), 9196-9216.
M. Mitoraj A. & Michalak, (2007). Journal of molecular modeling, 13(2), 347-355.
R. Zhong, A. Pöthig, Y. Feng, K. Riener, W. A. Herrmann, & F. E. Kühn, (2014). Green Chemistry, 16(12), 4955-4962.
R. Jagadeesan, G. Velmurugan & P. Venuvanalingam, (2015). RSC Advances, 5(98), 80661-80667.
Y. C. Chang, J. Y. Lee, C. F. Chiang, T. Y. Chung & H. M. Lee, (2025). ACS omega, 10(43), 52076-52088.
R. O. Pankov, D. O. Prima, A. Y. Kostyukovich, M. E. Minyaev & V. P. Ananikov, (2023). Dalton Transactions, 52(13), 4122-4135.
J. P. Perdew, K. Burke, & M. Ernzerhof, M. (1996). Physical Review Letters, 77(18), 3865–3868.
Salehzadeh, S., Gholiee, Y., & M. Bayat, (2013). Theoretical Study on the Alkali Cations 222 Cryptates in the Gas Phase and in Aqueous Solution
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Ehsan Alavipour (Author); Mehdi Bayat (Corresponding author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.



