Oxidative desulfurization of liquid hydrocarbon fuel: experimental and mathematical modeling study
Keywords:
Desulphurization, Fenton, dibenzothiophene, optimization, physical propertiesAbstract
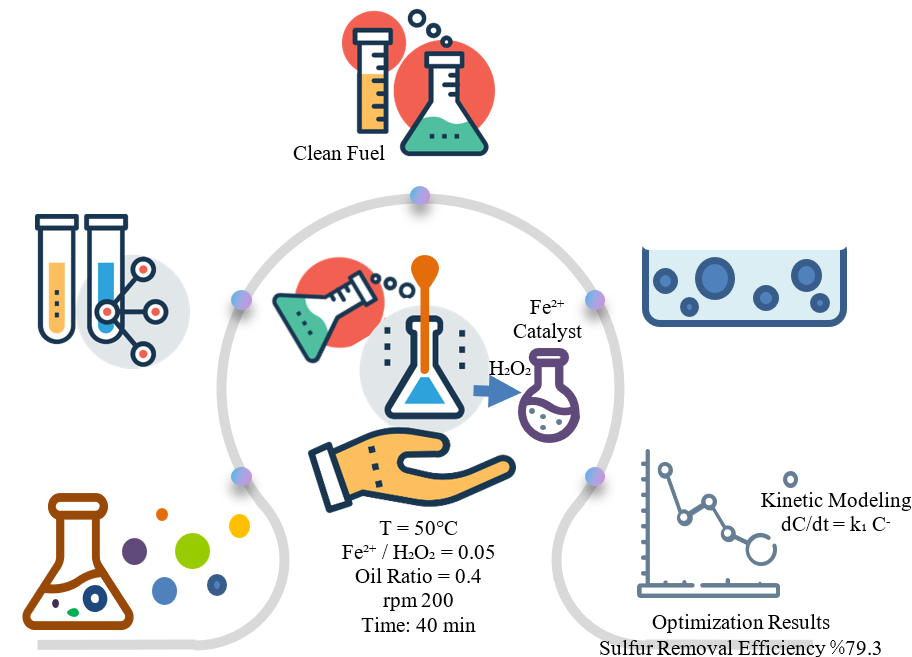
The positive increment trend in the world of continuously production of low sulfur content fuel as a transportation feed is under progressing. This issue is feasible by oxidative desulfurization as one of the most promising approaches. To cope with the difficulties and limitations of hydrotreating units, integration with oxidative desulfurization can be a vital solution. This assay presented Fenton process as a treatment method and explain a comprehensive analysis of different parameters and characteristics of this method on a model fuel of toluene. So contains experimental and modeling results for optimizing the reaction parameters to Fe+2/H2O2=0.05 molar ratio, T=50 °C, agitation speed= 200 rpm, oil volume ratio=0.4 and reaction time of 40 min under acidic pH. Then mass transfer characteristics estimated and process progressing studied. Since the oxidation occurred under estimated conditions, 79.3% efficiency achieved in the first-order kinetics. Parameters optimized for toluene as a model fuel nutriment by DBT as a sulfur contamination and oxidized under hydrogen peroxide as the oxidizing agent and ferrous ions as the catalyst. Which temperature and oil volume fraction play the most important roles in the progressing of the reaction. Kinetics modeling and calculations of reaction time constant, mass transfer coefficient, droplet diameter and surface area investigated under optimized condition
Downloads
References
CLM. Marcelis, M. van Leeuwen, HG. Polderman, AJH. Janssen & G. Lettinga, Biochem Eng J., 2003, 16(3), 253-264
MK. Bolla, HA. Choudhury & VS. Moholkar, Industrial & engineering chemistry research., 2012 , (1), 1-9.
A. Samokhvalov & BJ. Tatarchuk. Catalysis Reviews, (2015), 52(3), 381-410.
A. Deshpande, A. Bassi & Prakash, Energy and Fuels., 2005, 19(1), 28-34.
FA. Duarte, A. Mello & CA. Bizzi, et al., 2011, 90(6), 2158-2164.
AM. Dehkordi,MA. Sobati & MA. Nazem, J Chem Eng., 2009 17(5), 869-874.
M-C Lu, LCC. Biel LCC, M-W Wan, R. de Leon & S. Arco, Techniques Int J Green Energy., 2014, 11(8), 833-848.
M-Z. Sun, B. Zhang,Y-H. Wu, J. Zhu & D-Z. Zhao. Deep Oxidative Desulfurization of FCC Diesel Fuel with Ultrasound Pet Sci Technol, 2012, 30, 2471-2477.
Y. Dai, Y. Qi, D. Zhao & H. Zhang, An oxidative desulfurization method using ultrasound/Fenton’s reagent for obtaining low and/or ultra-low sulfur diesel fuel Fuel Process Technol .,2008, 89(10), 927-932.
Y. Dai, D. Zhao & Y. Qi. Sono-desulfurization oxidation reactivities of FCC diesel fuel in metal ion/H2O2 systems Ultrason Sonochem, 2011, 18(1), 264-268.
Z. Heidari, Study of Electro-Fenton Advanced Oxidation Process for Pentachlorophenol Removal in Aqueous Solutions Msc. thesis. 2015
P. De Filippis, M. Scarsella & N. Verdone, Ind Eng Chem Res., 2010, 49, 4594-4600.
S. V. Sambasivarao, Rate-limiting step analysis of the microbial desulfurization of dibenzothiophene in a model oil system NIH Public Access .,2015, 18(9), 1199-1216.
M. Massoudi, Int J Eng Sci., 2008, 46(7), 677-689.
M. Zhou & S. Kresta, Chemical Engineering Science Correlation of mean drop size and minimum drop size with the turbulence., 1998 53(11).
M. Raji-Asadabadi, H. Abolghasemi, MG. Maragheh & P. Davoodi-Nasab, Chem Eng Res Des., 2013 91(9), 1739-1747.
JO. Hinze, AIChE J., 1995, 1(3), 289-295.
CR.Wilke, P.Chang, AIChE J., 1995, 1, 264-270.
R. Gray, Interfacial Tension, 1991, 1-22. http://www.kruss.de/services/education-theory/glossary/interfacial-tension/.
R. Omrani manesh, Investigation of physico-chemical reatment of Shiraz landfill leachate Msc. thesis. 2015, 6(4), 417-429.
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Mahsa Jafari (Author); Mohammad Reza Khosravi-Nikou (Corresponding author); Mohsen Motavasel (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.



