Evaluating the Ion Adsorption Properties of CB3 and CN3 Nanoflakes for Nitrate and Nitrite Removal in Water Purification: A DFT Approach
-
Abstract
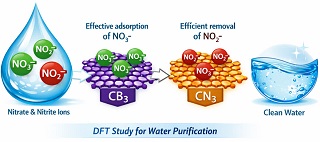
In this study, the adsorption and sensing performance of nitrate (NO₃⁻) and nitrite (NO₂⁻) ions on CB₃ and CN₃ nanoflakes were systematically investigated in both gas and aqueous environments. Density functional theory (DFT) and time-dependent DFT (TD-DFT) calculations were performed at the ωB97XD/6-31+G (d, p) level to optimize the geometries and evaluate the electronic, thermodynamic, and sensing-related properties of the NO₃⁻/NO₂⁻@CB₃ and NO₃⁻/NO₂⁻@CN₃ complexes. The calculated adsorption energies reveal that the CB₃–NO₂⁻ (model 2) complex exhibits the strongest interaction among all studied systems, indicating that CB₃ nanoflakes are highly effective adsorbents for nitrate and nitrite ions. The dipole moments and polarizabilities of the NO₃⁻@CB₃ and NO₃⁻@CN₃ complexes are significantly higher than those of their NO₂⁻ counterparts, suggesting greater sensitivity toward nitrate ions. Thermodynamic analyses confirm that the adsorption processes are exothermic and thermodynamically spontaneous, with enthalpy changes (ΔH) showing a slight decrease as the temperature increases from 298.15 to 328.15 K. The reduced energy gap (Egap) and chemical hardness (η) values for the adsorbed complexes indicate enhanced electrical conductivity and reactivity, supporting their suitability as sensitive sensors and efficient adsorbents. Further insights from atoms-in-molecules (AIM), electron localization function (ELF), and reduced density gradient (RDG) analyses demonstrate that the dominant interactions between NO₃⁻/NO₂⁻ ions and CB₃/CN₃ nanoflakes are governed by electrostatic forces and van der Waals interactions, with the NO₃⁻@CN₃ complex exhibiting the strongest interaction characteristics. Overall, the findings highlight CB₃ and CN₃ nanoflakes as promising candidates for the development of high-performance sensors and adsorbents for nitrate and nitrite ion removal from environmental systems.
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Mahdi Rezaei Sameti

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.



